
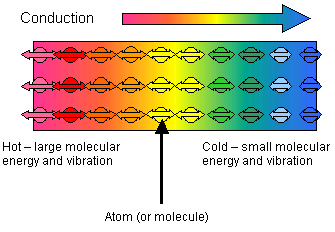
When one end of a solid rod is heated the atoms at that end vibrate – the more it is heated the more they vibrate.
This vibration is passed on from one atom to the next and so the energy is transferred down the rod and this is conduction.
In metals there are also a lot of free electrons and these play a big part in the energy transfer.
Some materials conduct heat better than others.
Metals conduct heat better than non-metals
Thick specimens conduct heat better than thin ones
Liquids are poor conductors of heat
Air is a very bad conductor of heat
The following list shows some common materials, starting with the one that conduct heat best and ending with the worst conductor:
silver sp copper sp aluminium s brass s iron s lead s rock s water s glass s rubber s air